Learn more about Aptar Pharma Expertise
in Nasal Drug Delivery
This Might Also Be of Interest
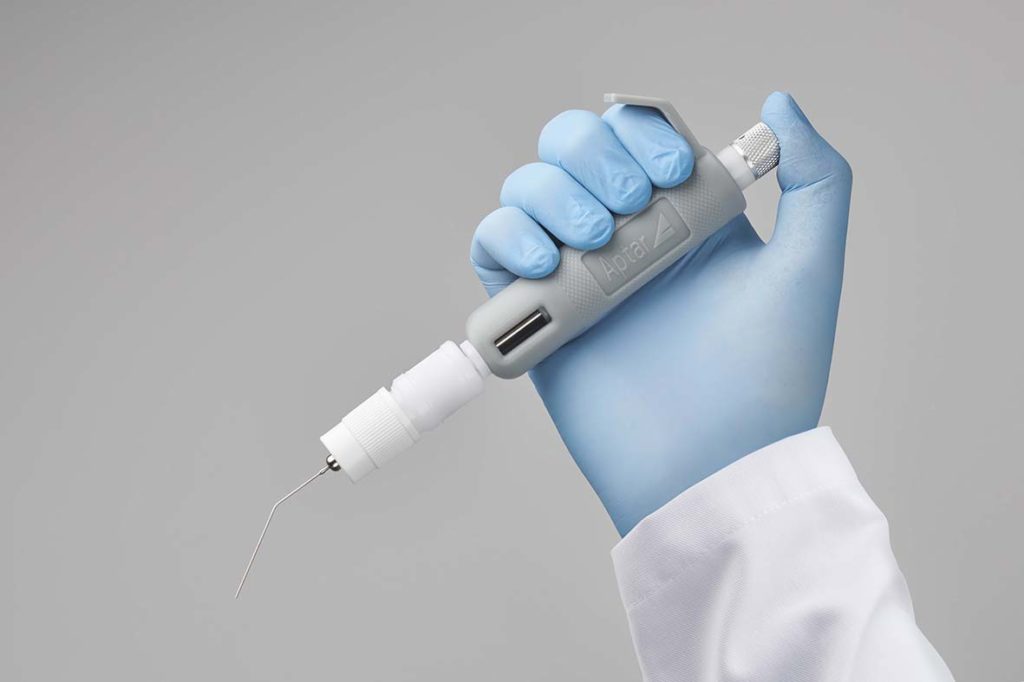
Case Study – Pre-Clinical Respiratory Research with PADA
Case Studies, Pharmaceutical, Innovation & Insights, Device Innovations, Product Solutions

Intranasal Immunization: Promises and Challenges
Webinars, Pharmaceutical, Sustainability, Innovation & Insights, Device Innovations, Market Insights, Product Solutions

Otimização de processos no desenvolvimento de produtos inalatórios orais e nasais.
Webinars, Pharmaceutical, Product Solutions
Using pharmacokinetics to confirm bioequivalence in fluticasone DPIs
Publications, Pharmaceutical, Innovation & Insights, Device Innovations, Product Solutions
